Advances in Environmental Protection
Vol.
09
No.
05
(
2019
), Article ID:
32424
,
8
pages
10.12677/AEP.2019.95090
Analytical Experiment and Mechanism Study of Surface Sediment on Fluorine Solution
Qing Liu1,2,3,4*, Junyao Yang5#, Yaqin Wang5, Liumei Ren5
1Shaanxi Provincial Land Engineering Construction Group Co. Ltd., Xi’an Shaanxi
2Institute of Land Engineering and Technology, Shaanxi Provincial Land Engineering Construction Group Co. Ltd., Xi’an Shaanxi
3Key Laboratory of Degraded and Unused Land Consolidation Engineering, The Ministry of Natural Resources, Xi’an Shaanxi
4Shaanxi Provincial Land Consolidation Engineering Technology Research Center, Xi’an Shaanxi
5College of Water Conservancy Science and Engineering, Taiyuan University of Technology, Taiyuan Shanxi

Received: Sep. 15th, 2019; accepted: Sep. 23rd, 2019; published: Sep. 30th, 2019

ABSTRACT
After the surface water is polluted, it reacts with the surface sediments and releases the pollutants to form the secondary pollution source. In order to study the adsorption and desorption of F− in soils and determine the chemical reactions that happened during the F− analysis, kinetic tests and adsorption and desorption experiments were used in this study. Combined with pseudo-second-order kinetic equation and isothermal adsorption equation, the reaction kinetics and adsorption analysis between the mud and the reservoir water show that when the reaction time and the initial concentration are changed; the reaction rate, the reaction process and the migration and transformation rules of F− in water are obtained. The results show that: 1) The kinetic process controlled by chemical reaction may be the dominant surface sediment, which can be described by pseudo second-order kinetic equation. The fitting equation is y = 0.7401x + 0.1266 and the correlation coefficient is R2 = 0.9985. 2) The isotherm of F− adsorption by surface sediment is in accordance with the linear relationship, and the saturated analytical volume of F− is 30.56 mg·g−1. 3) After the soil sample is in contact with the fluorine solution, the pH value of the solution increases with the increase of the F-mass concentration, indicating that OH− is continuously released in the F− analysis progresses; it can be seen from the change of other ions. The chemical reactions of F− occurring during the analysis process are precipitation-dissolution, adsorption-analysis, and ion exchange.
Keywords:Fluorine, Dynamics, Adsorption Analysis
表层沉积物对水中F−吸附及解析过程的影响
刘庆1,2,3,4*,杨军耀5#,王亚琴5,任柳妹5
1陕西省土地工程建设集团有限责任公司,陕西 西安
2陕西地建土地工程技术研究院有限责任公司,陕西 西安
3自然资源部退化及未利用土地整治工程重点实验室,陕西 西安
4陕西省土地整治工程技术研究中心,陕西 西安
5太原理工大学水利科学与工程学院,山西 太原

收稿日期:2019年9月15日;录用日期:2019年9月23日;发布日期:2019年9月30日

摘 要
地表水受污染后,与表层沉积物接触发生反应,污染物将释放出来,形成二次污染源。为了研究土壤对F−的解析作用,明确F−解析过程中发生的化学反应,本文采用动力学试验和吸附解吸试验,并结合伪二级动力学方程、等温吸附方程,模拟三泉水库底泥与库水之间的反应动力学过程和解析过程,得出当反应时间和初始浓度变化时,水中F−的反应速率,反应历程及其迁移转化规律。结果表明:1) 在表层沉积物上可能以化学反应控制的动力学过程为主,可以用伪二级动力学方程描述,拟合方程为t/qt = 0.7401t + 0.1266,相关系数R2 = 0.9985;2) 表层沉积物对F−的吸附解析等温线符合线性关系,拟合得出F−的饱和解析量为30.56 mg/g;3)土样与氟溶液接触后,溶液pH值随加入F−质量浓度增大而上升,说明随着F−解析的进行不断地有OH−被释放出来;从其他离子的变化可以看出,F−解析过程中发生的化学作用有沉淀–溶解,吸附–解析,离子交换作用。
关键词 :氟,动力学,吸附解析

Copyright © 2019 by author(s) and Hans Publishers Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


1. 引言
氟离子(F−)是重要的阴离子水体污染物,目前我国是世界上F−污染较为严重的国家之一 [1] 。土壤作为F−环境体系的枢纽,对F−在环境中的分布和运移具有直接影响 [2] 。土壤对F−的主要反应包含液膜扩散、表面吸附–解析、颗粒内部扩散过程等。
开展F−在土壤中的动态吸附与解吸特征研究,能帮助我们深入了解F−在水土体系中的迁移与转化机制。迄今为止,国内外对F−的吸附解吸已有很多的研究 [3] - [9] ,并针对不同的背景条件提出不同的等温吸附式,使用最多的有线性等温吸附式及非线性等温吸附式(包括Langmuir等温吸附方程、Freundlich经验式和Temkin方程式)。其中,王庆文等 [5] 通过研究土壤对矿井水中F−的吸附机理,得出土壤对F−的吸附符合Freundlich型吸附等温式;梁鹏等 [6] 的吸附试验表明MCLB和MCLRB对F−具有较高的吸附容量,吸附等温线Langmuir和Freundlich模型均能很好地拟合试验结果;阮建云等 [7] 认为氟的吸附率随氟浓度变化而变化,在高浓度下吸附的氟更易被解析,茶园土壤对F−的吸附可由Freundlich方程模拟,Langmuir方程只在低浓度(初始氟浓度0.5~2.0 mmol/L)时适用。Peek [8] 等对10个土壤中的氟化物的吸附和解析反应进行研究,认为Langmuir吸附等温式符合所有土壤的浓度范围,Freundlich的吸收等温线适用于初始F−浓度范围在0.0~2.5 mmol/l。Zhu [9] 等采用批处理技术,研究了酸性红土壤中对氟和磷的联合吸附,认为F−浓度的增加抑制了磷的吸收。纵观前人的研究,极少考虑到土壤对F−的吸附解吸相关化学反应机理。
本文采用三泉水库输水渠附近表层淤积层土壤,进行F−的动力学试验和静态吸附解析室内试验,研究了反应动力学过程、F−吸附解析特性及发生的化学反应机理,旨在从水文化学角度来揭示F−在地表沉积物中的累积、迁移转化规律,为土壤水体污染的治理提供理论支撑。
2. 材料与方法
2.1. 试验材料
供试土样:土壤于2016年8月13日取自山西省运城市新绛县三泉水库库中进水口附近,距表层0~10 cm。土样经去杂–风干–过筛处理,测得其物理化学性质详见表1~3。
Table 1. Particle analysis results and soil classification
表1. 颗粒分析结果与土的分类
注:采用国际制土壤质地分级标准。
Table 2. Physical characteristics of the test sample
表2. 供试土样物理特性
Table 3. Chemical characteristics of the test soil samples
表3. 供试土样化学特性
水样:试验配置8组水样,其中,F−浓度设置不同浓度(0.5 mg/L、1 mg/L、2 mg/L、4 mg/L、6 mg/L、8 mg/L、10 mg/L、12 mg/L),根据监测所采集的水库水样,得出水库中各离子的浓度,其他离子浓度按库水监测值配置。试验中其他离子的浓度均保持不变,分别为C( )=150 mg/L、C(Fe) = 0.4 mg/L、C(Mn) = 0.15 mg/L、C(Na+) = 120 mg/L、C(Ca2+) = 30 mg/L、C(Mg2+) = 60 mg/L。
2.2. 试验方法
动力学试验:对土样采用四分法各取120 g左右,每个土样各称取10份,每份10 g,分别置于250 ml的三角锥瓶中,根据测定的库水成分按照比例配置拟库水溶液。将土样和水样混合摇匀,在室温下以250 r/min的频率保持恒温水浴振荡,每间隔0.5、1、1.5、2、3、4、6、8、12、16、20和24 h取出一个样品,样品经离心-过滤-测量。
等温吸附试验:在室温环境下,取10.0 g过1 mm筛的土样置于已编号的锥形瓶中,加入配置的8种不同浓度的F离子溶液,pH = 7,在水浴振荡器中以250 r/min震荡8 h,使之达到吸附平衡,立刻取出在离心机中以5000 r/min离心8分钟后,过滤,取上清液,测定水样中相应离子的平衡浓度Ce(mg/L),计算单位质量土壤的吸附量Cs(mg/g) [10] 。
所有水样的测定均在室内水化学仪器实验室内完成,测定项目包括pH,氟离子、氯离子、硫酸根离子、铁、锰、钠离子、钙离子、镁离子共9项。其中pH采用雷磁ZDJ-4A自动电位滴定仪和pH复合电极测定,氟离子、氯离子、硫酸根离子采用883 Basic IC plus离子色谱仪测定,铁、锰、钠离子、钙离子、镁离子采用TAS-990原子吸收分光光度计测定。
3. 结果与分析
采用应用较广泛的伪一级动力学,伪二级动力学,颗粒内扩散,颗粒表面扩散,Elovich动力学五种动力学模型 [11] 模拟库水中各离子在底泥上的吸附过程,得到吸附动力学方程。
另,利用离子色谱仪对吸附前后的F−浓度进行测定,根据式(1)计算吸附量(Qe),以每克土样吸附F−的毫克数(mg/g)进行计算 [11] 。
(1)
式中,Qe为平衡时土壤对F−的吸附量(mg/g),C0为加入液中F−的浓度(mg/L),Ce为平衡时液体体积(L),m为加入的土样质量(g)。
3.1. 动力学试验结果分析
目前土壤化学动力学模型的研究主要有反应速率方程和吸附动力学方程。根据化学动力学原理和动力学实验结果,建立描述三泉水库库底泥中F−的长期释放特征与规律的模型,应用五种常用的动力学方程对F−反应过程进行拟合,拟合参数见表4。
Table 4. F− five kinds of adsorption kinetic model fitting
表4. F−五种吸附动力学模型拟合
综合分析拟合参数表,可以看出,五种动力学模型相关性均较好,相关系数达到显著性水平,伪二级动力学方程是最优模型,图1是伪二级动力学方程拟合情况。尽管采用经验公式拟合的结果并不能直接证明动力学机制,但模拟结果还是在一定程度上说明F−在水库底泥的吸附动态是不同过程共同作用的结果,并且伪二级动力学方程计算的平衡吸附量对实验随意误差的敏感性很小。因此认为沉积物的长期释放量规律性较强,可以用数学方程式t/qt = 0.74t + 0.1266表达。在土层浅的位置上可能以化学反应控制的动力学过程为主,可以用伪二级动力学方程描述。
3.2. F−解析情况
图2(a)是选用线性回归方程对溶液中F−的初始质量浓度与平衡后溶液F−的质量浓度之间的关系进行拟合,拟合的方程为:Qe = 0.0047Ce + 0.0115,相关决定系数R2达到0.99,达极显著相关水平,拟合结果可以看出,随着溶液中F-的增大,从土壤中解析出来的F−也逐渐增大,呈直线正相关水平,这与土中黏粒含量低有很大关系,黏粒具有较小的比表面,不宜吸附F−,且黏粒含量低的这些土质一般风化度较低,其氧化物含量较低,会减少对F−的吸附。根据测得的土样物理及化学性质可知,三泉水库进水口表层土样属粉沙,粘粒含量不高,其水溶性F−本底值为1.22 g·kg−1。

Figure 1. Pseudo-second-order kinetic fit
图1. 伪二级动力学拟合情况
此外,本文还选用非线性等温吸附方程Langmuir等温吸附方程、Freundlich经验式和Temkin方程式对试验数据进行拟合分析。图2(b)~(d)是得到的拟合结果,根据相关系数R2由好到差进行排列:Langmuir 方程式
3.3. 解析机理分析
3.3.1. 环境(pH)变化
图3表示的是随着F−浓度的增大,溶液pH的变化趋势,从图中可以看出,随着F-浓度的增大,pH值也随之增大,最终稳定在7.45~7.5。随着F−加入浓度的增加,吸附平衡液的pH值上升,说明随着F−解析的进行不断地有OH−被释放出来。因OH−半径与F−的半径非常接近,它们的电荷数相等,极化度极相似,因此随着氟的解析不断地有OH−被释放出来,导致土壤溶液pH逐渐上升。
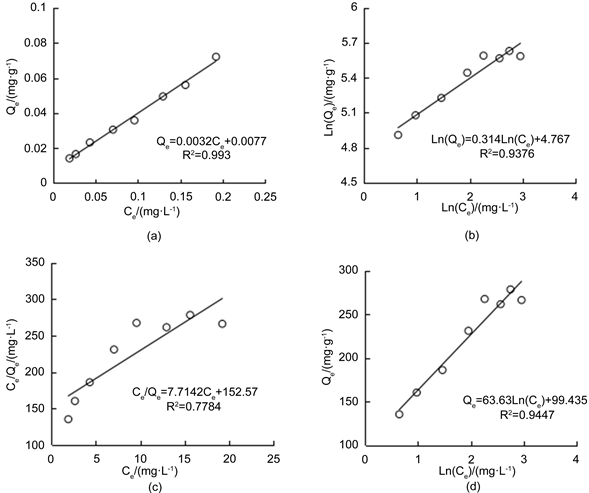
Figure 2. The fitting curve of F− desorption
图2. F−解析拟合曲线

Figure 3. The change of pH value with F− concentration
图3. pH值随F−离子浓度变化规律
3.3.2. 其他离子的变化
图4是随着F−浓度变化其他离子的吸附解吸情况,F−的吸附解析常伴随有其它离子的解吸 [12] ,这种吸附解析本质上是竞争吸附。从图中可以看出,试验过程中,部分离子发生解析反应,部分离子发生吸附反应。其中Fe、Na+、 发生解析反应, 解析量最大,Na+次之;Mn、Ca2+、Mg2+发生吸附反应,其中Ca2+、Mg2+吸附量相对较大。岩土中各种化学物质与水溶液的相互作用不断影响水溶液的酸碱条件和氧化还原条件,促进或抑制水溶液中各种化学反应 [12] [13] [14] 。
从图4中可以看出,其他离子随着F−浓度的变化而变化,除铁离子外,铁离子解析量始终保持在0.0015 mg/g (图4(a))。从图4(b)可以看出,锰离子随F-浓度的增大呈线性增大,同时发现,在F−浓度相对较低的情况下,锰离子的吸附量逐渐减小,直至C(F−) = 10 mg/L时,结果表示,锰开始从土中解析出来。从图4(c)~(f),Na+、Mg2+、Ca2+、 变化规律比较一致,但Na+、 是从土向水发生解析反应,而Mg2+、Ca2+是从水向土发生吸附反应,它们的变化规律大致相同,在低浓度时逐渐增大,C(F−) = 4~6 mg/L时,浓度变化出现峰值,随后降低,最终趋于稳定。其中Na+浓度在C(F−) = 4~6 mg/L解析量达到0.393 mg/g,随后稳定在0.38 mg/g左右;Mg2+浓度在C(F−) = 6 mg/L时吸附量达到−0.2295 mg/g,随后稳定在−0.232 mg/g左右。
 (a) Fe浓度变化
(a) Fe浓度变化
 (b) Mn浓度变化
(b) Mn浓度变化
 (c) Na+浓度变化
(c) Na+浓度变化
 (d) Ca2+浓度变化
(d) Ca2+浓度变化
 (e) Mg2+浓度变化
(e) Mg2+浓度变化
 (f)
SO42-
浓度变化
(f)
SO42-
浓度变化
Figure 4. Other ions with the F− concentration trend
图4. 其他离子随F−浓度变化趋势图
3.3.3. 吸附机理分析
分析试验数据说明,水体中的F−有明显的累积性,属于迁移累积元素。该土样对F−的解析主要是通过羟基交换反应而达到解析F−的效果,可能F−参与络合反应取代羟基的结果。该土样在F−浓度低时解析速率高,其可能原因是在低浓度时,土样表面活性位点较多,Mg2+、Ca2+很容易到达吸附位点并解析到土样表面;随着F−浓度提高,土样表面活性位点越来越少,且吸附到土样表面的Mg2+、Ca2+与溶液中的F−存在静电斥力作用,减弱土样对F−的解析,从而降低了F−的解析速率。在微酸和偏碱性条件下,F−易于与金属离子形成络合物,还可能与溶液中的钙和镁等离子形成沉淀,不利于氟的迁移而有利于F−的富集 [12] 。
由土壤胶体具有巨大的比表面积和表面能的特性可知,土壤胶体表面可以通过静电吸附与溶液中的离子进行交换反应,按照吸附强度的不同,土壤在溶液中进行交换吸附反应时分为专性吸附与非专性吸附 [5] 。氟化物在水中以F−的形式出现,其来源主要是萤石(CaF2)、磷灰石(Ca(Cl, F, OH)(PO4)3)等矿物的风化水解。氟化物能与水中的一些阳离子如Al3+、Fe3+形成络合物。溶解的氟化物能与Ca2+反应,产生CaF2,因此Ca2+的浓度是控制F−存在多少的一个重要因素 [15] 。
4. 结论
在表层沉积物上可能以化学反应控制的动力学过程为主,可以用伪二级动力学方程描述,拟合方程为t/qt = 0.7401t + 0.1266,相关系数R2 = 0.9985。该土样对F−的吸附等温线符合线性方程,拟合得出对F−的饱和吸附量为30.56 mg/g。此外,土解析出F−后的平衡溶液pH值随F−加入质量浓度增大而上升,对土样中F−在水土界面迁移转化响较大的是沉淀–溶解和吸附–解吸平衡。
研究还发现,三泉水库库水中的F−浓度本身并不高,但通过表面淤积层后会解析出一部分F−,从而造成F−二次污染。由于湖泊主要处于地下水的补给区,入渗补给作用强烈,应深入研究库水入渗对地下水质的影响,以便改善生态环境。
文章引用
刘 庆,杨军耀,王亚琴,任柳妹. 表层沉积物对水中F-吸附及解析过程的影响
Analytical Experiment and Mechanism Study of Surface Sediment on Fluorine Solution[J]. 环境保护前沿, 2019, 09(05): 669-676. https://doi.org/10.12677/AEP.2019.95090
参考文献
- 1. 曹春, 康宏亮, 李萍萍, 等. 聚丙烯偕胺肟改性羊毛对氟离子的吸附及机理[J]. 高分子学报, 2016(4): 486-493.
- 2. 荆秀艳, 袁周燕, 杨红斌, 等. 土氟静态吸附特性及其影响因素[J]. 生态环境学报, 2008, 17(5): 1818-1821.
- 3. 杨军耀. 水–土系统氟迁移影响因素分析[J]. 工程勘察, 1998(3): 42-44.
- 4. 江霜英, 高廷耀. 粘土对水中氟离子吸附去除机理的研究[J]. 化工环保, 2003, 23(4): 204-208.
- 5. 王庆文. 土壤对矿井水中F−的吸附动力学和吸附机理初探[J]. 河南理工大学学报(自然科学版), 2009, 28(6): 807-810.
- 6. 梁鹏. 稀土改性壳聚糖树脂的制备、表征及对氟离子的吸附特性研究[D]: [博士学位论文]. 青岛: 中国海洋大学, 2013.
- 7. 阮建云, 马立锋, 石元值, 等. 茶园土壤对氟的吸附与解吸特性[J]. 茶叶科学, 2001, 21(2): 161-165.
- 8. Peek, D.C. and Volk, V.V. (1985) Fluoride Sorption and Desorption in Soils. Soil Science Society of America Journal, 49, 583-586. https://doi.org/10.2136/sssaj1985.03615995004900030010x
- 9. Zhu, M.X., Ding, K.Y., Jiang, X. and Wang, H.-H. (2007) Investigation on Co-Sorption and Desorption of Fluoride and Phosphate in a Red Soil of China. Water Air & Soil Pol-lution, 183, 455-465. https://doi.org/10.1007/s11270-007-9394-0
- 10. 陈男. 天然及合成多孔性粘土材料对地下水中氟化物的吸附性能研究[D]: [博士学位论文]. 北京: 中国地质大学, 2012.
- 11. 韩德刚, 高盘良. 化学动力学基础[M]. 北京: 北京大学出版社, 1987.
- 12. 董岁明. 氟在土–水系统中的迁移机理与含氟水的处理研究[D]: [博士学位论文]. 西安: 长安大学, 2004.
- 13. 梁秀娟. 吉林西部洋沙泡水库水土环境中氟的迁移转化机理研究[D]: [博士学位论文]. 长春: 吉林大学, 2008.
- 14. Nur, T., Loganathan, P., Nguyen, T.C., et al. (2014) Batch and Column Adsorption and Desorption of Fluoride Using Hydrous Ferric Oxide: Solution Chemistry and Modeling. Chemical Engineering Journal, 247, 93-102. https://doi.org/10.1016/j.cej.2014.03.009
- 15. 王洪涛. 多孔介质污染物迁移动力学[M]. 北京: 高等教育出版社, 2008.
NOTES
*第一作者。
#通讯作者。
